Abstract
Introduction The severity of coronavirus disease 2019 (COVID-19) is associated with the development of a cytokine storm driven by classical monocytes. Activation of the NLRP3 inflammasome mediates the production of IL-1β - a key pro-inflammatory cytokine that in excessive quantities causes tissue injury.
Most CLL patients have an impaired humoral response to vaccination with subsequent higher risk of complications from COVID-19 infection. Currently, no pre-exposure test exists to assess the risk of a dysregulated immune response to COVID-19. We previously reported that open reading frame 8 (ORF8), a SARS-CoV-2 protein, when glycosylated and secreted into the blood, stimulates CD14+ monocytes and activates the NLRP3 pathway to produce IL-1β (Ruan et al, AAI 2022).
We hypothesized that functional characteristics of CLL patients’ monocytes may predispose patients to develop severe COVID-19. First, we developed the ORF8 monocyte stimulation test (OMST), which measures monocyte reactivity via flow cytometry after stimulation of patient monocytes ex vivo with ORF8. Secondly, we applied the OMST to CLL patients to learn about its association with severe COVID-19 infection.
Methods We identified patients from the Mayo Clinic CLL Database who were diagnosed with COVID-19 infection via a positive SARS-CoV-2 RT-PCR nasal swab between July 2020 to February 2022 and had a cryopreserved pre-COVID-19 peripheral blood mononuclear sample (PBMC) available. Our study was approved by the IRB, and samples were stored in the Mayo Clinic Predolin Biobank.
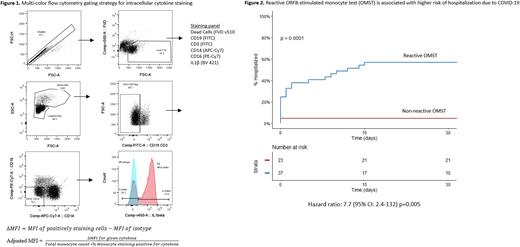
For the OMST, samples were thawed and either stimulated with ORF8 or unstimulated (control) for 24 hours. The samples were harvested and intracellular cytokine/chemokine levels (IL-1β, IL-8, and CCL-2) in monocytes were analyzed by flow cytometry using the depicted gating strategy and formulas to calculate delta MFI (dMFI) and adjusted MFI (Figure 1). Clinical outcomes were abstracted by chart review. Severe infection was defined as hospitalization due to COVID-19 infection. Fisher's exact test, Wilcoxon rank sum test, and matched paired analysis were used to compare categorical, continuous, and paired variables, respectively. Receiver-operating curves (ROC) were used to calculate the area under the curve (AUC), sensitivity, specificity, and optimal cutoff levels for a reactive OMST. The optimal cutoff point was chosen as the point with the highest Youden index. Time to severe COVID-19 infection was calculated using the Kaplan-Meier method and associations of factors with severe COVID-19 were assessed using Cox proportional hazard model (CPHM).
Results 60 patients were identified. The median age at COVID-19 diagnosis was 74 years (63-80). The median number of COVID-19 vaccinations before infection was 0 (range 0-3). The median time interval between vaccination and infection was 106 days (2-322). At diagnosis, 25 (42%) patients were receiving CLL-directed therapy and 22 (37%) were on a BTK-inhibitor. 22 patients had severe COVID-19 and 6 patients died.
The OMST demonstrated that CD14+ monocytes had a significantly higher dMFI for IL-1β, IL-8, and CCL-2 after ORF8 stimulation compared to controls (495 vs. 158, p<0.0001), (298 vs. 121, p<0.001), and (385 vs. 171, p <0.0001) respectively. By ROC analysis, the AUC for IL-1β adjusted MFI was 0.68, 0.67 for IL-8 adjusted MFI, and 0.37 for CCL-2 adjusted MFI. A reactive OMST was defined by ROC as an IL-1β adjusted MFI >= 0.18 (sensitivity 67%, specificity 75%). The median time to hospitalization after COVID-19 infection for patients with a reactive OMST was 12 days (95% CI: 2 - not reached) versus not reached (NR) for patients with a non-reactive OMST [95% CI: NR-NR] with a hazard ratio of 7.7 (95% CI: 2.4-132, p=0.005) [Figure 2]. The following variables were not significant on univariate CPHM: age, gender, IGHV mutation, CLL-IPI risk, Charlson Comorbidity score, smoking status, number of mRNA COVID-19 vaccinations, receipt of monoclonal antibodies, or on therapy for CLL.
Conclusions We have developed a novel assay, the OMST, that measures the reactivity of human monocytes to ORF8 and has significant value in determining the severity of COVID-19 infection in CLL patients. These findings are independent of patient age, sex, comorbidity score, prognostic models, and vaccination status. The OMST has the potential to assess monocyte reactivity and its implications in a broad range of clinical situations.
Disclosures
Kay:AbbVie: Membership on an entity's Board of Directors or advisory committees, Research Funding; AstraZeneca: Membership on an entity's Board of Directors or advisory committees, Other: Data Safety Monitoring Committee; Beigene: Membership on an entity's Board of Directors or advisory committees; Behring: Membership on an entity's Board of Directors or advisory committees; Cytomx Therapy: Membership on an entity's Board of Directors or advisory committees, Other: Data Safety Monitoring Committee; Dava Oncology: Membership on an entity's Board of Directors or advisory committees; Janssen: Membership on an entity's Board of Directors or advisory committees, Other: Data Safety Monitoring Committee; Juno Therapeutics: Membership on an entity's Board of Directors or advisory committees; Oncotracker: Membership on an entity's Board of Directors or advisory committees; Pharmacyclics: Membership on an entity's Board of Directors or advisory committees, Research Funding; Targeted Oncology: Membership on an entity's Board of Directors or advisory committees; BMS: Other: Data Safety Monitoring Committee, Research Funding; Dren Bio: Other: Data Safety Monitoring Committee; Morpho-sys: Other: Data Safety Monitoring Committee; Rigel: Other: Data Safety Monitoring Committee; Celgene: Other: Data Safety Monitoring Committee, Research Funding; Genentech: Research Funding; MEI Pharma: Research Funding; Sunesis: Research Funding; TG Therapeutics: Research Funding; Tolero Pharmaceuticals: Research Funding. Parikh:Merck: Consultancy, Research Funding; GlaxoSmithKline: Consultancy; Genetech: Consultancy; Ascentage Pharma: Research Funding; AbbVie: Consultancy, Research Funding; Janssen: Research Funding; AstraZeneca: Consultancy, Research Funding; TG Therapeutics: Research Funding; Phamacyclics: Consultancy, Research Funding; Adaptive Biotehcnologies: Consultancy. Witzig:Curio Science: Honoraria; ADC Therapeutics: Membership on an entity's Board of Directors or advisory committees; Karyopharm: Other: Clinical Trail Support; Kura Oncology: Other: Clinical Trail Support.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal